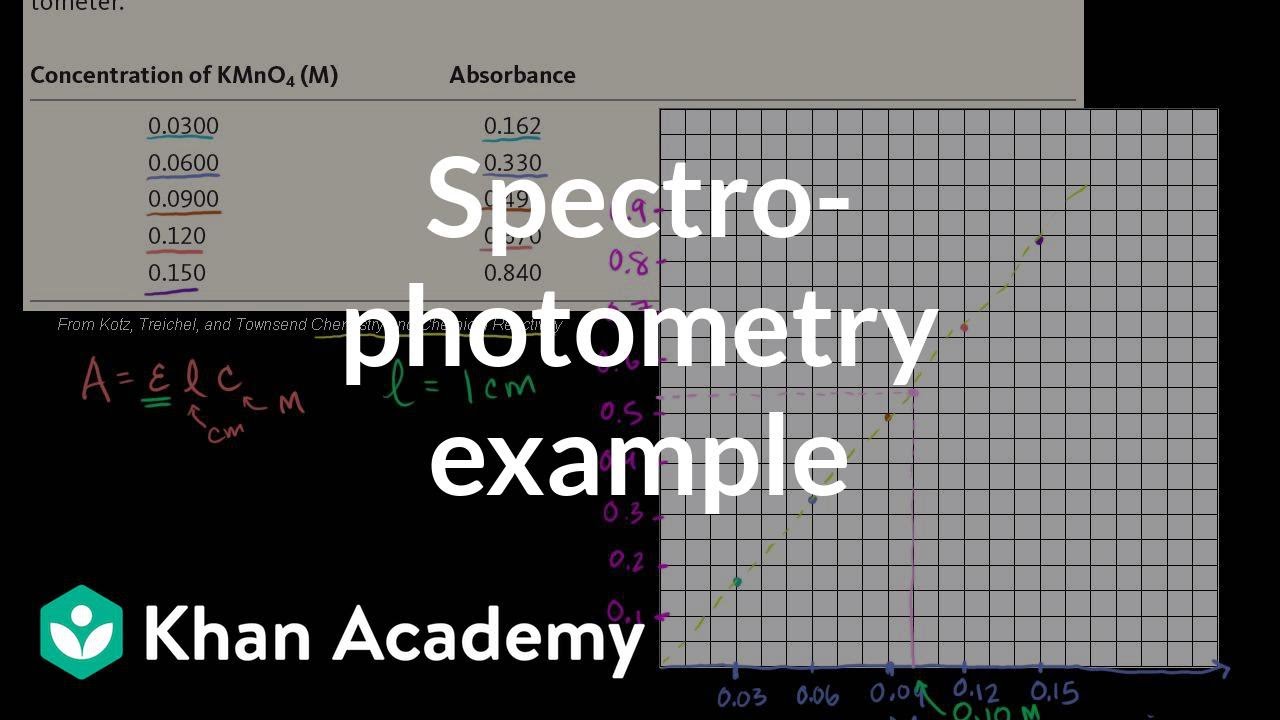
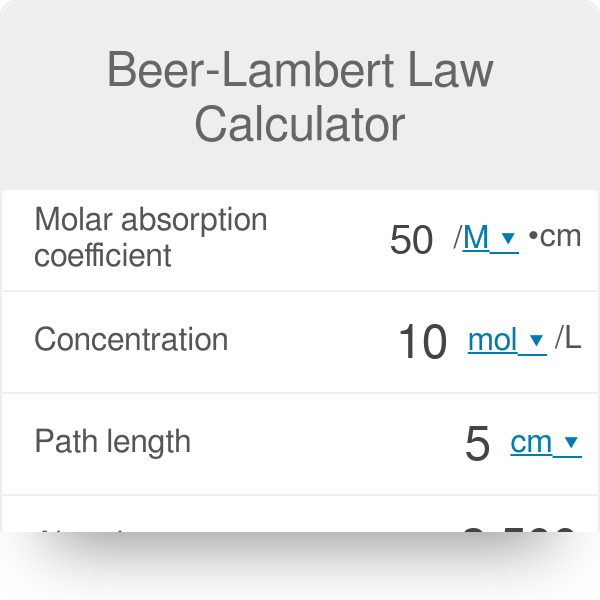
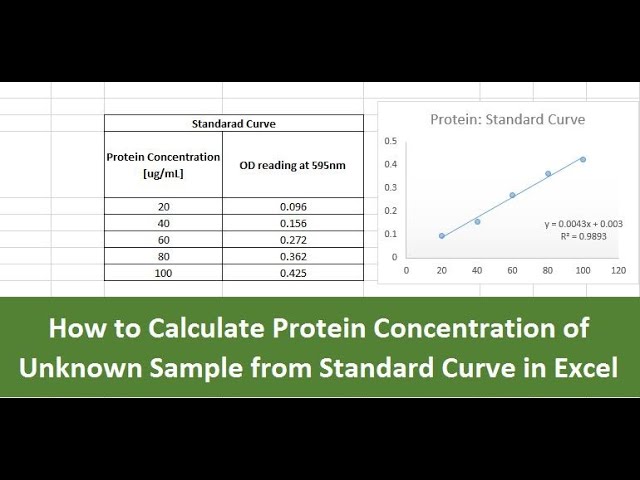
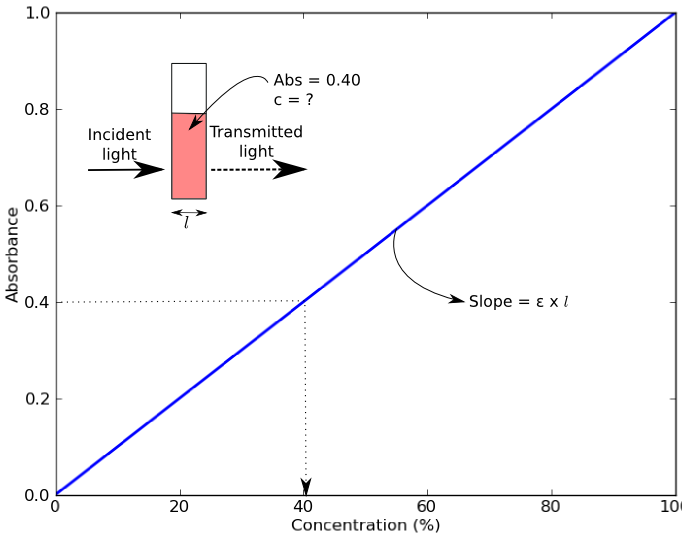
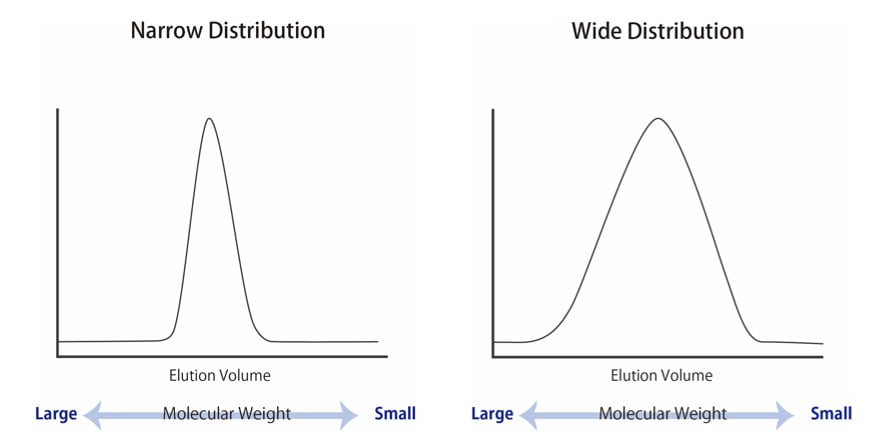
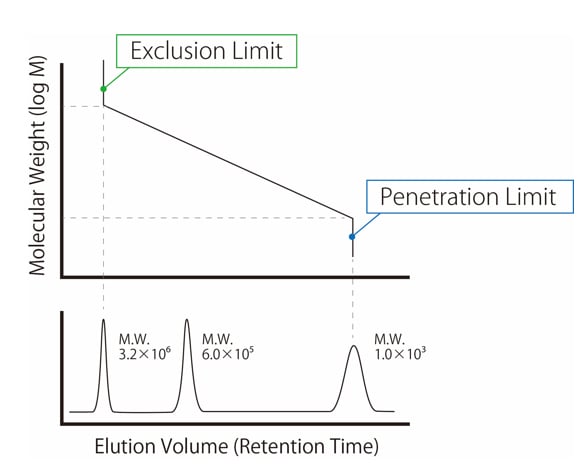
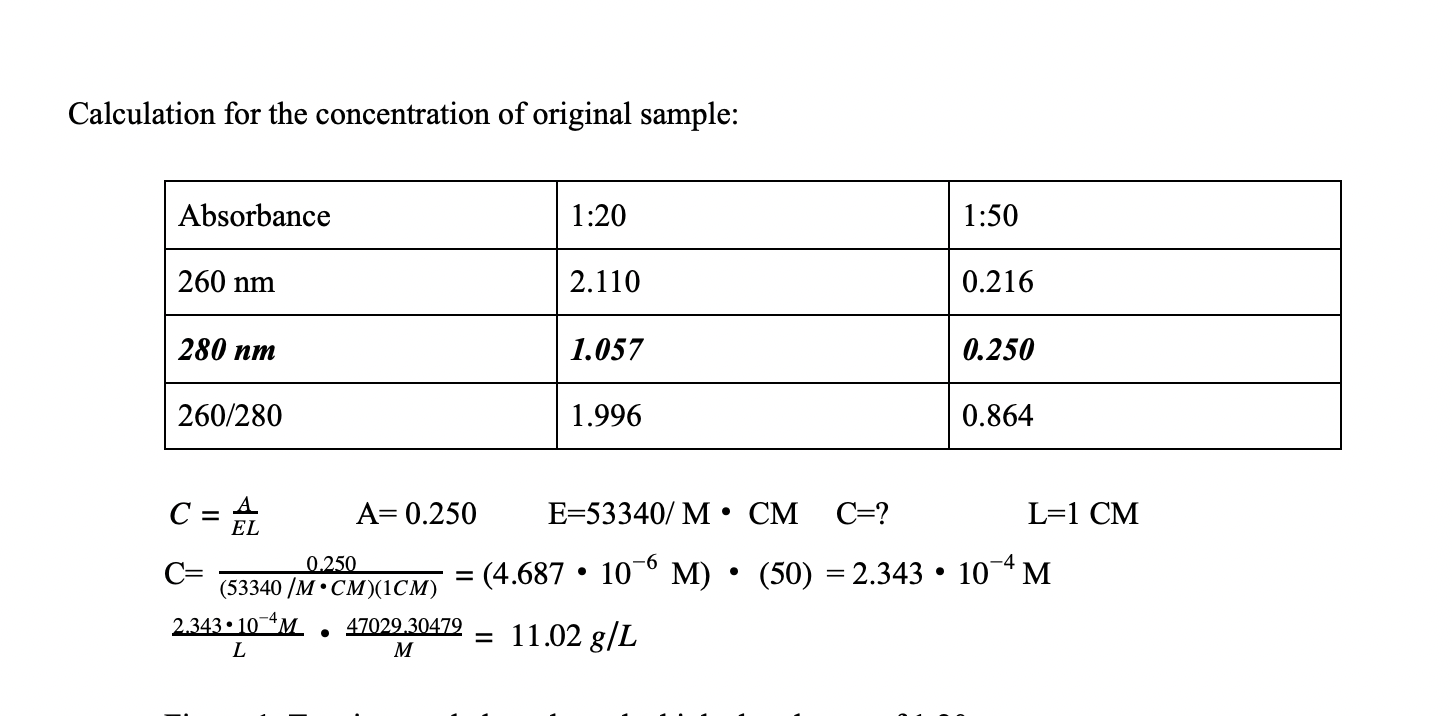
SOLVED: Molar Absorptivity Absorbance is directly proportional to the path length, b, and the concentration, C of the absorbing species. Beer's Law states that A = cl From Spectra A is the
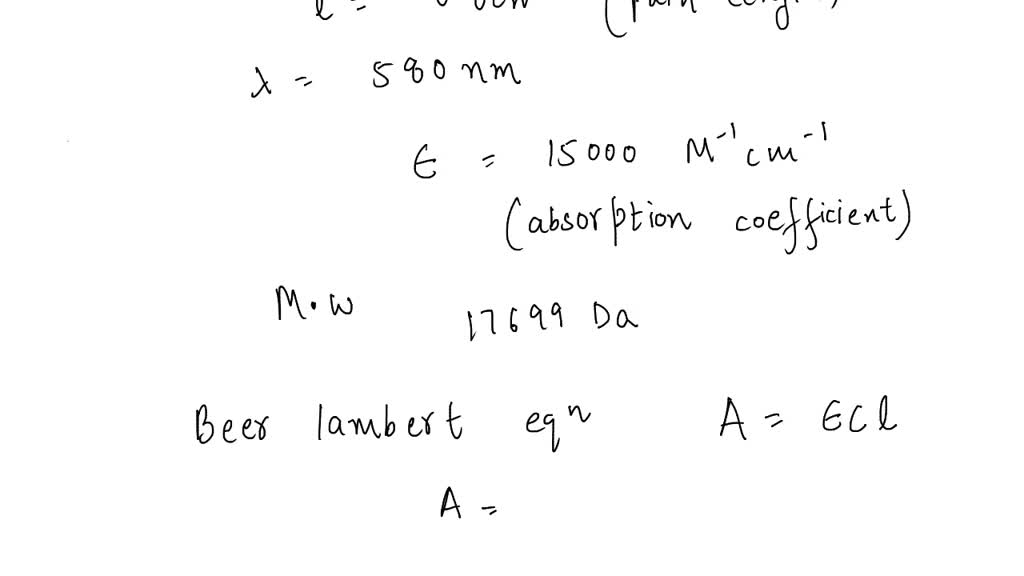
SOLVED: The absorption coefficient of myoglobin at 580 nm is 15,000 M-1cm-1. What is the absorbance of 1 mg/ml solutionin a cuvette with a 0.8 cm path length? Molecular weight of myoglobin

Molecular Weight Effect on the Absorption, Charge Carrier Mobility, and Photovoltaic Performance of an Indacenodiselenophene-Based Ladder-Type Polymer | Chemistry of Materials
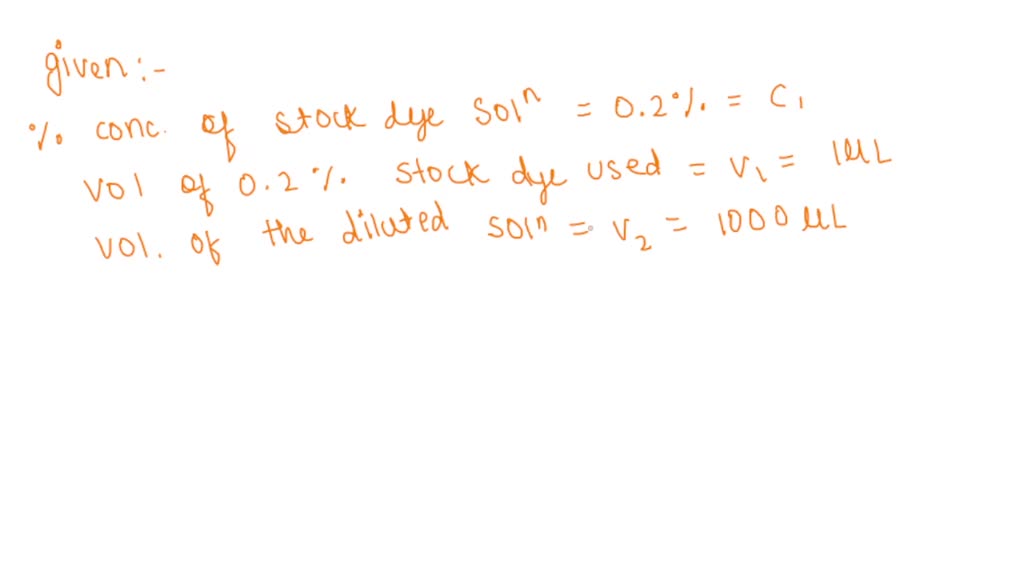
SOLVED: Use Beer's law to convert the given absorbance to molar concentration, then to percent concentration. e = 97000 M-1cm-1 Molecular weight = 792.8 grams/mol To calculate percent error, you will compare