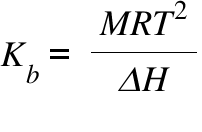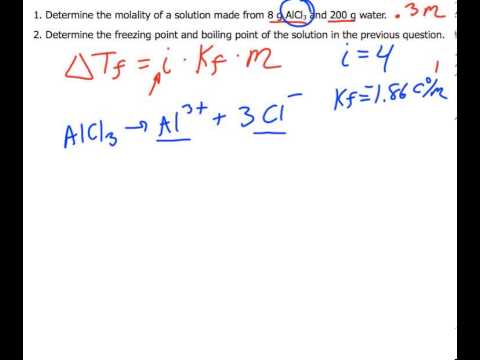Boiling Point Elevation and Freezing Point depression - Overview ( Video ) | Chemistry | CK-12 Foundation

Calculation the boiling point of a 1M aqueous solution (density 1.04 g mL^-1 )of potassium chloride (Kb for water = 0.52 K kg mol^-1 , Atomic masses: K = 39u, Cl =