
Calculate the hydrogen ion concentration in the following biological fluids whose pH are given below:(a) Human muscle - fluid, 6.83 (b) Human stomach fluid, 1.2 (c) Human blood, 7.38 (d) Human saliva, 6.4.

SOLVED: Calculate the concentration of an aqueous HI solution that has pH=2.50 . HI is a strong acid.
![SOLVED:MATHEMATICAL Calculate the hydrogen ion concentration, [H^+] for each of the following materials: (a) Saliva, pH 6.5 (b) Intracellular fluid of liver, pH 6.9 (c) Tomato juice, pH 4.3 (d) Grapefruit juice, SOLVED:MATHEMATICAL Calculate the hydrogen ion concentration, [H^+] for each of the following materials: (a) Saliva, pH 6.5 (b) Intracellular fluid of liver, pH 6.9 (c) Tomato juice, pH 4.3 (d) Grapefruit juice,](https://cdn.numerade.com/previews/c92ef7f1-c83f-4027-9f05-6b11ebe1b21e_large.jpg)
SOLVED:MATHEMATICAL Calculate the hydrogen ion concentration, [H^+] for each of the following materials: (a) Saliva, pH 6.5 (b) Intracellular fluid of liver, pH 6.9 (c) Tomato juice, pH 4.3 (d) Grapefruit juice,

Calculating Hydronium ion concentration from pH (and identifying molecules based on pH) : r/chemhelp

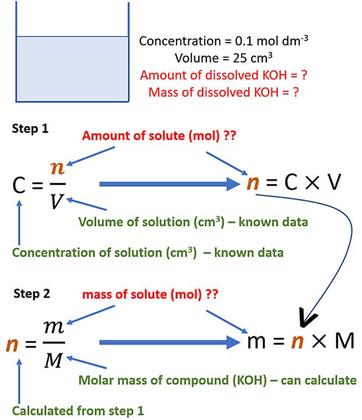

![Calculating [OH-], pH and pOH from Kb Calculating [OH-], pH and pOH from Kb](https://www.mi.mun.ca/users/pfisher/chemistry1011_135/img007.gif)

![Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE - YouTube Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/gn1CgBzShps/maxresdefault.jpg)




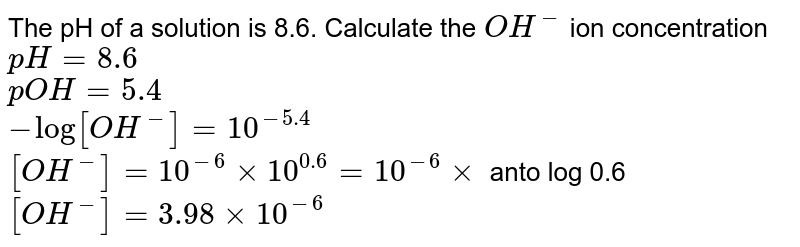



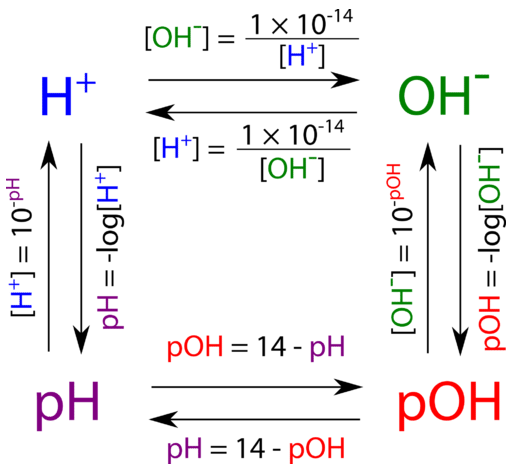



![Given [H+] or [OH-], Calculate pH & pOH - YouTube Given [H+] or [OH-], Calculate pH & pOH - YouTube](https://i.ytimg.com/vi/ghIYaqo0Ycc/maxresdefault.jpg)